Borane Complexes: BH3•LIn addition, borane rapidly reduces aldehydes, ketones, and alkenes.
Keeping this in consideration, what can BH3 not reduce?
The main use of boranes is for the hydroboration of alkenes, one of the most versatile and useful reactions in organic synthesis. Borane also reduces aldehydes, ketones, lactones, epoxides, acids, tertiary amides, and nitriles but does not reduce esters.
Similarly, what can be reduced to aldehyde? Carboxylic acids, esters, and acid halides can be reduced to either aldehydes or a step further to primary alcohols, depending on the strength of the reducing agent; aldehydes and ketones can be reduced respectively to primary and secondary alcohols.
Subsequently, one may also ask, what does BH3 reduce?
BH3·L (borane complexes)
Reduce carboxylic acids in the presence of esters, amides and halides.
Can BH3 reduce amines?
The initial product of an amide reduction with BH3 is the amine-BH3 complex which has to be decomposed to liberate the free amine product. Borane reagents generally show better functional group tolerance than the reactive Aluminium hydride reagents, but will reduce alkenes, acids, esters etc.
Related Question Answers
Is BH3 a strong reducing agent?
Doen the group (↓) i.e, from N to Bi, the atomic size increases, hence the bond length A-H increases consequently A-H bond strength decreases and tendency to behave as reducing agent increases i.e., BiH3 is the strongest reducing agent amongst all the hydrides of group 15 elements. How do carboxylic acids reduce to aldehydes?
There are no known general methods of reducing carboxylic acids to aldehydes, though this can be done indirectly by first converting the acid to the acyl chloride and then reducing the chloride. Why is BH3 a reducing agent?
Since,the BiH3 is the least stable (because the size of central atom is greatest & therefore its tendency to form stable covalent bond with small hydrogen atom decreases,as a result the bond strength decreases) in this series,hence the reducing character increases. Does LiAlH4 reduce acetals?
The reduction of cyclic acetals and ketals derived from aromatic or aliphatic aldehydes and ketones with CpTiCl3-LiAlH4 system in diethyl ether at 30°C affords the corresponding hydroxyethyl ethers and the corresponding alkyl benzene or aliphatic hydrocarbons. Can LiAlH4 reduce ketones?
LiAlH4 is a strong, unselective reducing agent for polar double bonds, most easily thought of as a source of H-. It will reduce aldehydes, ketones, esters, carboxylic acid chlorides, carboxylic acids and even carboxylate salts to alcohols. Amides and nitriles are reduced to amines. Can H2 reduce carboxylic acid?
H2/Pt will reduce aldehydes and ketones, but not carboxylic acids. Does Lindlar's catalyst reduce alkenes?
Lindlar's catalyst is a palladium catalyst poisoned with traces of lead and quinoline, that reduce its activity such that it can only reduce alkynes, not alkenes. Does borane react with water?
While ammonia borane does not react with water, partially and fully thermally reacted AB does react to produce hydrogen. Use of a small amount of water (50 mL) with a large quantity of hydrogen storage material (>100 g) can result in very high reaction temperatures. Is LDA a reducing agent?
Lithium Diisopropylamide (LDA) as an Efficient Reducing Agent for Thioketones—Mechanistic Consideration. Treatment of thiocarbonyl compounds with excess of lithium diisopropylamide (LDA) leads to corresponding thiols or sulfides depending on the work-up procedure. What is BH3 used for?
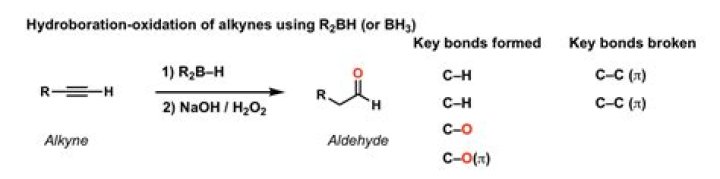
The most important use of BH3 is for the hydroboration of alkenes and alkynes. For the reaction of alkenes, there are two important things to keep in mind. The reaction is selective for the less substituted alcohol (regioselective). This is usually referred to as “anti-Markovnikoff†selective. How do you reduce an ester to an aldehyde?
Esters can be converted to aldehydes using diisobutylaluminum hydride (DIBAH). The reaction is usually carried out at -78 oC to prevent reaction with the aldehyde product. Are Hemiacetals stable?
Only a few hemiacetals are stableLike their hydrates, the hemiacetals of most ketones (sometimes called hemiketals) are even less sta- ble than those of aldehydes.
Can lah reduce conjugated alkenes?
LiAlH4 is a rather hard nucleophilic reductant (HSAB Principle) which means it reacts with electrophiles, and alkenes are not electrophiles. But LAH has a tendency to reduce the double bond if it's in conjugated system. Why is LiAlH4 a good reducing agent?
Because aluminium is less electronegative than boron, the Al-H bond in LiAlH4 is more polar, thereby, making LiAlH4 a stronger reducing agent. Addition of a hydride anion (H:–) to an aldehyde or ketone gives an alkoxide anion, which on protonation yields the corresponding alcohol. What is borane made of?
borane, any of a homologous series of inorganic compounds of boron and hydrogen or their derivatives. What will reduce an aldehyde to a primary alcohol?
Aldehydes can be reduced to primary alcohols (RCHO → RCH2OH) with many reducing agents, the most commonly used being lithium aluminum hydride (LiAlH4), sodium borohydride (NaBH4), or hydrogen (H2) in the presence of a transition catalyst such as nickel (Ni), palladium (Pd), platinum (Pt), or rhodium (Rh). What is reduced when an aldehyde is oxidized?
Aldehydes reduce the complexed copper(II) ion to copper(I) oxide. Because the solution is alkaline, the aldehyde itself is oxidised to a salt of the corresponding carboxylic acid. Can you reduce alcohols?
Nucleophiles that displace tosylate groups include hydride ions, H-. Therefore, tosylates formed from alcohols undergo nucleophilic substitution reacctions with hydride sources, such as lithium aluminum hydride (LiAlH4, aka LAH). The net result of the process is the reduction of alcohols to alkanes. What products are formed when aldehydes and ketones are reduced?
Aldehydes produce 1º-alcohols and ketones produce 2º-alcohols. In metal hydrides reductions the resulting alkoxide salts are insoluble and need to be hydrolyzed (with care) before the alcohol product can be isolated. Which reagent reduce aldehyde and ketone to alcohol?
Formation of alcohol from aldehydes or ketones requires either of the particular reducing agents. The reducing reagents are sodium borohydride (NaBH4 ) or lithium aluminium hydride (LiAlH4). How do you remove a carbonyl?
Aldehydes and ketones may also be reduced by hydride transfer from alkoxide salts. The reductive conversion of a carbonyl group to a methylene group requires complete removal of the oxygen, and is called deoxygenation. How do you turn an alcohol into an aldehyde?
Hydride reacts with the carbonyl group, C=O, in aldehydes or ketones to give alcohols. The substituents on the carbonyl dictate the nature of the product alcohol. Reduction of methanal (formaldehyde) gives methanol. Reduction of other aldehydes gives primary alcohols. Can H2 NI reduce aldehydes?
Functional groups such as acid halide, cyanide, nitro, aldehyde, ketone, alkene, and alkyne are easily reduced by H2/Ni. How is aldehyde oxidised?
What is formed when aldehydes are oxidized? It depends on whether the reaction is done under acidic or alkaline conditions. Under acidic conditions, the aldehyde is oxidized to a carboxylic acid. Under alkaline conditions, this couldn't form because it would react with the alkali. Why Dibal H reduces esters to aldehydes?
of DIBAL-H gives aldehyde . This is because the transfer of one hydride to the carbonyl carbon of ester ,leads to the formation of the tetrahedral intermediate stable at low temperature. Does H2 Raney nickel reduce esters?
Reduce aldehyde or ketone to alcohol: use H2 with Raney nickel iv. It will not reduce an acid or an ester. ii. LiAlH4 will reduce an aldehyde, ketone, acid, or ester to the corresponding alcohol. Does DIBAL react with aldehydes?
What it's used for: DIBAL is a strong, bulky reducing agent. Unlike lithium aluminum hydride, it will not reduce the aldehyde further if only one equivalent is added. It will also reduce other carbonyl compounds such as amides, aldehydes, ketones, and nitriles. What does BH3 THF do?
Borane–tetrahydrofuran is a dipolar bond charge-transfer complex composed of borane and tetrahydrofuran (THF). These solutions are used for reductions and hydroboration, reactions that are useful in synthesis of organic compounds. Does lah reduce amides?
* LiAlH4 reagent can reduce aldehydes to primary alcohols, ketones to secondary alcohols, carboxylic acids and esters to primary alcohols, amides and nitriles to amines, epoxides to alcohols and lactones to diols.