Polymer crystallinity can be determined with DSC by quantifying the heat associated with melting (fusion) of the polymer. This heat is reported as Percent Crystallinity by normalizing the observed heat of fusion to that of a 100 % crystalline sample of the same polymer. Besides, how is degree of crystallinity measured?
The specific enthalpy of fusion of a sample determined from the peak area is proportional to its degree of crystallinity. The proportionality factor, Δhc, is equal to the enthalpy of fusion of a 100% crystalline material. If the value is known, the degree of crystallinity can be determined from the melting curve.
Furthermore, what is degree crystallization? Crystallinity refers to the degree of structural order of a solid. In a crystal, the arrangement of atoms or molecules is consistent and repetitive. Many materials such as glass ceramics and some polymers can be prepared in such a way as to produce a mixture of crystalline and amorphous regions.
Considering this, what is degree of crystallinity polymer?
The fraction of the ordered molecules in polymer is characterized by the degree of crystallinity, which typically ranges between 10% and 80%. Higher values are only achieved in materials having small molecules, which are usually brittle, or in samples stored for long time at temperatures just under the melting point.
What is crystallinity index?
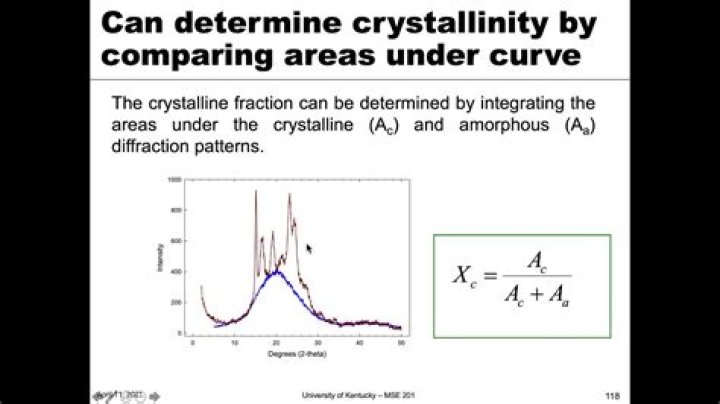
Crystallinity index (CI) is a quantitative indicator of crystallinity. Various techniques, such as X-ray. diffraction (XRD), Fourier transform infrared spectroscopy (FTIR) and Raman spectroscopy, and many. methods based on these techniques have been used to define the CI of HA.
Related Question Answers
How do you get crystallinity?
Polymer crystallinity can be determined with DSC by quantifying the heat associated with melting (fusion) of the polymer. This heat is reported as Percent Crystallinity by normalizing the observed heat of fusion to that of a 100 % crystalline sample of the same polymer. Why is crystallinity important?
For semi-crystalline polymers, the degree of crystallinity is one of its most important physical parameters since it reflects the sample's morphology and determines various mechanical properties, such as the Young modulus, yield stress as well as the impact strength. How is crystallinity calculated from DSC?
Polymer crystallinity can be determined with DSC by quantifying the heat associated with melting (fusion) of the polymer. This heat is reported as Percent Crystallinity by normalizing the observed heat of fusion to that of a 100 % crystalline sample of the same polymer. How does the degree of crystallinity affect a polymer?
Crystallinity defines the degree of long-range order in a material, and strongly affects its properties. The more crystalline a polymer, the more regularly aligned its chains. Increasing the degree of crystallinity increases hardness and density. How does DSC work?
DSC – Differential Scanning Calorimetry is a technique in which the difference in the amount of heat required to increase the temperature of a sample and reference are measured as a function of temperature. Both the sample and reference are maintained at nearly the same temperature throughout the experiment. How do you increase the crystallinity of a polymer?
Increase in concentration which can occur via solvent evaporation, induces interaction between molecular chains and a possible crystallization as in the crystallization from the melt. Crystallization from solution may result in the highest degree of polymer crystallinity. Why would we want to synthesize a polymer with a high degree of crystallinity?
Crystallinity defines the degree of long-range order in a material, and strongly affects its properties. The more crystalline a polymer, the more regularly aligned its chains. Increasing the degree of crystallinity increases hardness and density. What is meant by crystallinity?
Crystallinity. Crystallinity refers to the degree of structural order in a solid. In a crystal, the atoms or molecules are arranged in a regular, periodic manner. The degree of crystallinity has a big influence on hardness, density, transparency and diffusion. How do you find the crystallinity index from the XRD graph?
After subtracting the diffractogram of the amorphous cellulose from the diffractogram of the whole sample, the CI was calculated by dividing the remaining diffractogram area due to crystalline cellulose by the total area of the original diffractogram. X-ray diffraction spectra of amorphous cellulose examples. How do you find the density of a polymer?
Once the volume of the
polymer is known, its
density can be
calculated.
Density by Pycnometry
- mp = mass of polymer.
- mf = mass of dry empty flask.
- mfw = mass of flask filled with water.
- mfwp = mass of flask filled with polymer and water.
How does branching affect crystallinity?
Short branches interfere with the formation of crystals, that is, they reduce the amount of crystallinity whereas long branches undergo side chain crystallization because they are able to form lamellar crystals of their own. Which polymer is more likely to crystallize?
Linear polymers are more likely to crystallize that branched ones. In addition, polymers having isotactic structures will normally have a higher degree of crystallinity that those having atactic structures. How does crystallinity affect toughness?
Crystallinity makes a material strong, but it also makes it brittle. A completely crystalline polymer would be too brittle to be used as plastic. The amorphous regions give a polymer toughness, that is, the ability to bend without breaking and the ability to absorb impact energy. Can we have a 100% crystalline polymer Why or why not?
Remember we said that many polymers contain lots of crystalline material and lots of amorphous material. Almost no polymer is 100% crystalline and, in fact, most polymers are only around 10-30% crystalline. There's a way we can find out how much of a polymer sample is amorphous and how much is crystalline. How does Tacticity affect polymer properties?
The tacticity of a polymer is determined by what side of the polymer chain the pendant groups are on. This relative position can have dramatic effects on the physical properties of the polymer. If the monomer adds where the pendant group adds to the opposite side of the polymer backbone, it is called syndiotactic. What does it mean when a polymer is crosslinked?
Polymer chemistry. Crosslinking is the general term for the process of forming covalent bonds or relatively short sequences of chemical bonds to join two polymer chains together. Why does crystallinity improve the strength of a polymer?
Crystallinity: The crystallinity of the polymer increases strength, because in the crystalline phase, the intermolecular bonding is more significant. Hence, the polymer deformation can result in the higher strength leading to oriented chains. What makes a polymer transparent?
Seeing clearly through polymers A material is transparent if it lets light rays through without deflection or scatter. In a semi-crystalline polymer, by contrast, the thousands of crystallites that form its regular structure diffract light rays, making it opaque. What is a semi crystalline material?
Semi-crystalline materials have a highly ordered molecular structure with sharp melt points. They do not gradually soften with a temperature increase, instead, semi-crystalline materials remain solid until a given quantity of heat is absorbed and then rapidly change into a low viscosity liquid. How do you increase your crystallinity?
Crystallinity is usually induced by cooling a melt or a dilute solution below its melting point. The later can result in the growth of single crystals. Crystallization can also be induced by stretching a polymer. What does degree of polymerization mean?
The degree of polymerization, or DP, is the number of monomeric units in a macromolecule or polymer or oligomer molecule. What does high crystallinity mean?
Crystallinity defines the degree of long-range order in a material, and strongly affects its properties. The more crystalline a polymer, the more regularly aligned its chains. Increasing the degree of crystallinity increases hardness and density. Is PE amorphous or crystalline?
Those regions are therefore neither crystalline nor amorphous and are classified as semicrystalline. Examples of semi-crystalline polymers are linear polyethylene (PE), polyethylene terephthalate (PET), polytetrafluoroethylene (PTFE) or isotactic polypropylene (PP). Is PC amorphous or crystalline?
Amorphous thermoplastics are generally used at temperatures below their Tg, where they can be brittle, just like glass. There are, however, certain exceptions. Polycarbonate (PC) is amorphous yet it is considered tough at temperatures well below its Tg. What are mechanical properties of polymers?
The physical properties (such as transition temperature, viscosity, etc.) and mechan- ical properties (such as strength, stiffness, and toughness) depend on the molecular weight of polymer. The lower the molecular weight, lower the transition temperature, viscosity, and the mechanical properties. Why are amorphous polymers brittle?
Thermal and mechanical properties. Below their glass transition temperature, amorphous polymers are usually hard and brittle because of the low mobility of their molecules. Relatively strong intermolecular forces in semicrystalline polymers prevent softening even above the glass transition temperature. What is another word for crystalline?
Synonyms. crystal clear transparent limpid lucid pellucid clear.