What are the types of optical isomerism?
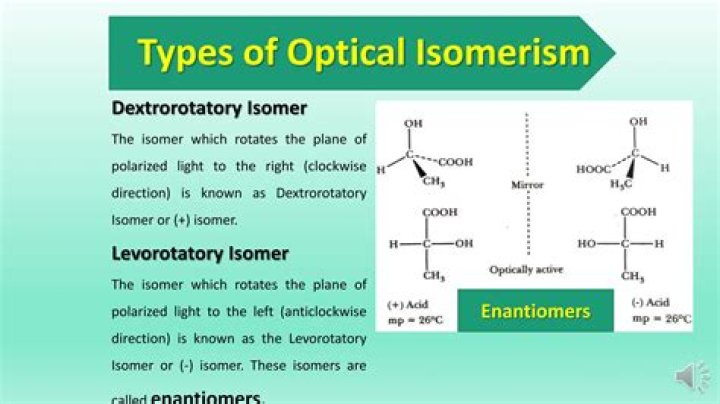
Thereof, what is optical isomerism?
Optical isomers are two compounds which contain the same number and kinds of atoms, and bonds (i.e., the connectivity between atoms is the same), and different spatial arrangements of the atoms, but which have non-superimposable mirror images. Each non-superimposable mirror image structure is called an enantiomer.
Similarly, what is Stereoisomerism and its types? Stereoisomers are a type of isomer where the order of the atoms in the two molecules is the same but their arrangement in space is different. The two main types of stereoisomerism are: Diastereomerism (including 'cis-trans isomerism') Optical Isomerism (also known as 'enantiomerism' and 'chirality')
Thereof, what are types of isomerism?
Isomers do not necessarily share similar chemical or physical properties. Two main forms of isomerism are structural or constitutional isomerism, in which bonds between the atoms differ; and stereoisomerism or spatial isomerism, in which the bonds are the same but the relative positions of the atoms differ.
How do you identify an optical isomer?
The mirror image method uses a mirror image of the molecule to determined whether optical isomers exist or not. If the mirror image can be rotated in such a way that it looks identical to the original molecule, then the molecule is said to be superimposable and has no optical isomers.
Related Question Answers
What is optical isomerism explain with example?
Optical isomers are compounds that are nonsuperimposable mirror images of each other. If the arrangement in space makes the two isomers nonsuperimposable mirror images of each other, we call them optical isomers or enantiomers. An example is the amino acid alanine.What causes optical isomerism?
Optical isomers can occur when there is an asymmetric carbon atom. An asymmetric carbon atom is one which is bonded to four different groups. It forms a chiral centre of the molecule. The four groups can be something hideously complex, or something comfortably simple like a hydrogen atom or a chlorine atom.How many types of optical isomers are there?
Simple substances which show optical isomerism exist as two isomers known as enantiomers. A solution of one enantiomer rotates the plane of polarisation in a clockwise direction. This enantiomer is known as the (+) form.Why is optical isomerism important in drug design?
Drug isomerism has opened a new era of drug development. Currently, knowledge of isomerism has helped us in introducing safer and more effective drug alternatives of the newer as well as existing drugs. Many existing drugs have gone chiral switch i.e., switching from racemic mixture to one of its isomers.What are D and L stereoisomers?
D and L enantiomersEditD and L enantiomers refer to the configurational stereochemistry of the molecule. L isomers have the hydroxy group attached to the left side of the asymmetric carbon furthest from the carbonyl, while D isomers have the hydroxy group on the right side.
What is D and L nomenclature?
The d/l system (named after Latin dexter and laevus, right and left) names molecules by relating them to the molecule glyceraldehyde. One example is the chiral amino acid alanine, which has two optical isomers, and they are labeled according to which isomer of glyceraldehyde they come from.Are all optical isomers optically active?
Optical isomers also have no axis of symmetry, which means that there is no line that bisects the compound such that the left half is a mirror image of the right half. Optical activity is the interaction of these enantiomers with plane-polarized light.What is isomerism give example?
Isomers are functional isomers when the two or more compounds have an identical molecular formula but differ in the functional group present. For example, we can represent C3H6O as a ketone and as an aldehyde.What are the two types of isomerism?
There are two general types of isomers. Constitutional isomers are molecules of different connectivity—analogous to simple bracelets in which the order of red and green beads is different. The second type is stereoisomers. In stereoisomers the connectivity is the same, but the parts are oriented differently in space.What is structural isomerism give examples?
Structural isomers are molecules with the same molecular formula, but their atoms have different arrangements or bonds. for example : Butane and isobutane have the same number of carbon (C) atoms and hydrogen (H) atoms, so their molecular formulas are the same. How satisfied are you with the answer?What are the three types of isomers?
There are three types of structural isomers: chain isomers, functional group isomers and positional isomers. Chain isomers have the same molecular formula but different arrangements or branches. Functional group isomers have the same formula but different functional groups.What is Tautomerism with example?
Tautomers are isomers of a compound which differ only in the position of the protons and electrons. The carbon skeleton of the compound is unchanged. A reaction which involves simple proton transfer in an intramolecular fashion is called a tautomerism.What causes chirality?
The feature that is most often the cause of chirality in molecules is the presence of an asymmetric carbon atom. In chemistry, chirality usually refers to molecules. Two mirror images of a chiral molecule are called enantiomers or optical isomers.What is a Stereoisomer example?
Geometric isomers (also called cis/trans isomers) are a type of stereoisomer resulting from a double bond or a ring structure. For example, oleic acid is the common name given to the molecular on the left whose chemical formula is C18H34O2 and which has a cis oriented double bond between carbons 9 and 10.What does Epimer mean?
Epimer: One of a pair of stereoisomers that differ in the absolute configuration of a single stereocenter. When the molecule has only one stereocenter then the epimers are enantiomers. When the molecule has two or more stereocenters then the epimers are diastereomers.What does enantiomer mean?
Enantiomers are chiral molecules that are mirror images of one another. Furthermore, the molecules are non-superimposable on one another. This means that the molecules cannot be placed on top of one another and give the same molecule. Chiral molecules with one or more stereocenters can be enantiomers.How does Stereoisomerism arise?
Geometric isomerism is one form of stereoisomerism. These isomers occur where you have restricted rotation somewhere in a molecule. Think about what happens in molecules where there is unrestricted rotation about carbon bonds - in other words where the carbon-carbon bonds are all single.How many stereoisomers are there?
With 1 chiral center, there are 2 isomers, 2 chiral centers, 4 possible isomers, 3 centers, 8 isomers and 4 centers, 16 possible stereoisomers. For an arbitrary number (n) of chiral centers in a molecule there are as many as 2n possible stereoisomers. Sucrose, with nine chiral carbons, has 29 stereoisomers, or 512.Are diastereomers mirror images?
Diastereomers are stereoisomers that are not related as object and mirror image and are not enantiomers. Unlike enatiomers which are mirror images of each other and non-sumperimposable, diastereomers are not mirror images of each other and non-superimposable.What defines a Stereocenter?
Stereocenter (chiral center): An atom with three or more different attachments, interchanging of two of these attachments leads to another stereoisomer. Most commonly, but not limited to, an sp3 (tetrahedral) carbon atom bearing four different attachments.Are all optical isomers enantiomers?
Optical isomers are Non Superimposable Mirror Images of each other; a set of optical isomers are called enantiomers. Enantiomers can not be interconverted without breaking bonds. They have mostly identical physical properties and can often only be told apart by what we call Optical Activity.Which of the following compounds can show optical isomerism?
The compound [Cr(C2O4)3]3−shows optical isomerism because its mirror image is not superimposable. It is the complex of the type [M(AA)3]n±. Three symmetrical bidenate ligands (oxalate ligands) are attached to Cr atom. The complex is optically active and exists in the form of d and l isomers.Which does not show optical isomerism?
Solution : optical isomerism Here, aa represents bidentate ligand, x or y represents monodentate ligand and M represents central metal ion. Hence, [Co(NH3)3Cl3]0 due to presence of symmetry elements does not exhibit optical isomerism.What causes optical activity?
Theory. Optical activity occurs due to molecules dissolved in a fluid or due to the fluid itself only if the molecules are one of two (or more) stereoisomers; this is known as an enantiomer.How do you calculate the number of optical isomers?
Hence, we have derived that, if 'n' (number of chiral centers) is odd for a compound with similar ends, then:- Number of meso isomers=2(n−1)/2.
- Total number of optical isomers=2n−1.
- Number of enantiomers=2n−1−2(n−1)/2.