What does ape in ape man stand for?
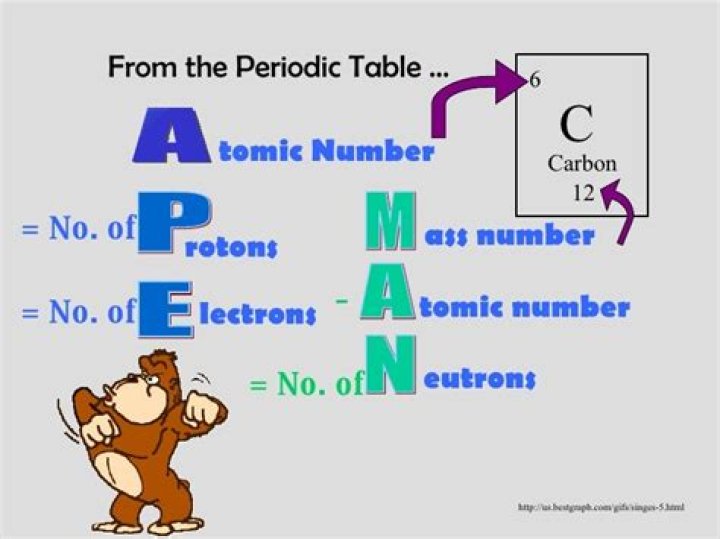
Accordingly, what does the acronym ape man stand for?
Protons, Neutrons and Electrons
Beside above, what is meant by atomic mass? The atomic mass of a single atom is simply its total mass and is typically expressed in atomic mass units or amu. By definition, an atom of carbon with six neutrons, carbon-12, has an atomic mass of 12 amu.
Accordingly, what were the main characteristics of ape man?
These creatures had small brain-sizes like apes, and large premolar and molar teeth, yet their canine teeth were small and they walked bipedally. They are regarded as hominids. These African early hominids were called by Raymond Dart Australopithecus.
What does the atomic number represent?
The atomic number is the number of protons in the nucleus of an atom. The number of protons define the identity of an element (i.e., an element with 6 protons is a carbon atom, no matter how many neutrons may be present).
Related Question Answers
What is Ape Man used for?
Description. An acronym used to help students remember the math to determine the protons, neutrons and electrons that make up an atom. There are two per page and can be cut and glued/taped into a composition journal.What is an valence electron?
Valence electrons are the electrons in the outermost shell, or energy level, of an atom. For example, oxygen has six valence electrons, two in the 2s subshell and four in the 2p subshell.What is ape science?
"Ape" has been used as a synonym for "monkey" or for naming any primate with a human-like appearance, particularly those without a tail. Biologists have traditionally used the term "ape" to mean a member of the superfamily Hominoidea other than humans, but more recently to mean all members of Hominoidea.How many electrons does oxygen have?
2, 6Where is most of the mass in an atom?
nucleusWhat charge does an electron have?
As summarized in Table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. The negative charge of one electron balances the positive charge of one proton. Both protons and neutrons have a mass of 1, while electrons have almost no mass.What is the definition of subatomic particles?
Definition - Subatomic particles. Subatomic particles: particles which are smaller than atoms, such as protons, electrons and neutrons.How many protons does sodium have?
11Who was the first ape man?
The earliest hominin, of presumably primitive bipedalism, is considered to be either Sahelanthropus or Orrorin, both of which arose some 6 to 7 million years ago.Who is apeman?
Apeman or ape-man may refer to: historically, a term for archaic humans, see: Missing link (human evolution) Pithecanthropus ("ape-man"), historical taxon now synonymous with Homo.Why can't you see an atom with the naked eye?
Atoms are really small. So small, in fact, that it's impossible to see one with the naked eye, even with the most powerful of microscopes. At least, that used to be true. Now, a photograph shows a single atom floating in an electric field, and it's large enough to see without any kind of microscope.How is atomic mass calculated?
For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called the mass number. This is because each proton and each neutron weigh one atomic mass unit (amu). By adding together the number of protons and neutrons and multiplying by 1 amu, you can calculate the mass of the atom.Who invented electron?
Joseph John ThomsonWhat do you call the element from atomic number 57 71?
Lanthanides. What do you call the element series from atomic number 57-71? Silicon.How do atoms exist?
Atoms exist in free states in the form of molecule. Molecules may be formed by the combination of atoms of two or more different elements. For example molecule of water. It is formed by the combination of two atoms of hydrogen and one atom of oxygen.What is the average atomic mass?
The average atomic mass (sometimes called atomic weight) of an element is the weighted average mass of the atoms in a naturally occurring sample of the element. Average masses are generally expressed in unified atomic mass units (u), where 1 u is equal to exactly one-twelfth the mass of a neutral atom of carbon-12.What is absolute mass?
In an atom most of the space is vacant, thereby electrons, protons, and neutrons constitute the mass of the atom. Thus, the absolute mass of an atom is the mass of its subatomic particles i.e., the mass of the electrons + protons + neutrons.What is difference between atomic mass and atomic weight?
Atomic weight is the weighted average of the naturally occurring isotopes. So again, the mnemonic for memorizing the difference between atomic mass and atomic weight is: atomic mass is the mass of an atom, whereas atomic weight is the weighted average of the naturally occurring isotopes.What is the difference between atomic mass and gram atomic mass?
When atomic mass is defined in terms of gram it is known as gram atomic mass. When molecular mass is defined in terms of gram it is known as gram molecular mass. ExplanationAtomic weight if oxygen is 16amu. 1 mole of oxygen is 6.02×10^23 therefore 1 amu =1.661×10^-24gram.What 3 things does the atomic number tell you?
The three main atomic particles are protons, neutrons and electrons. The atomic number of an atom identifies the number of protons in the atom. This is the defining characteristic of an element.What are the first 30 elements?
The first 30 elements of the periodic table and their symbols| A | B |
|---|---|
| Lithium | Li |
| Beryllium | Be |
| Boron | B |
| Carbon | C |
What did you notice on the elements atomic number?
The atomic number uniquely identifies a chemical element. It is identical to the charge number of the nucleus. In an uncharged atom, the atomic number is also equal to the number of electrons. The sum of the atomic number Z and the number of neutrons N gives the mass number A of an atom.What is the atomic number of oxygen?
8Why is oxygen atomic number 8?
The Periodic Table of elements can tell you a lot about the elements. Oxygen with the symbol O has the atomic number 8 which means it is the 8th element in the table. The number eight also means that oxygen has eight protons in the nucleus. Therefore oxygen has 8 electrons.What is the first 20 element?
First 20 Elements| Atomic Number | Element | Symbol |
|---|---|---|
| 17 | Chlorine | Cl |
| 18 | Argon | Ar |
| 19 | Potassium | K |
| 20 | Calcium | Ca |