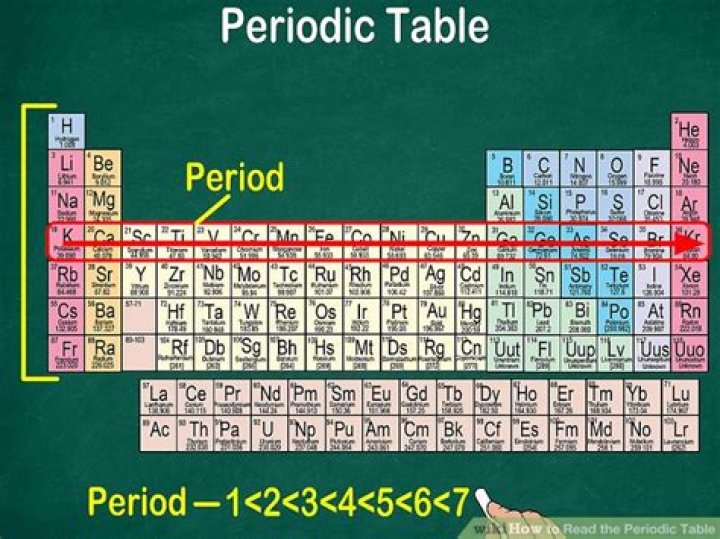
When you look at the periodic table, each row is called a period (Get it? Like PERIODic table.). All of the elements in a period have the same number of atomic orbitals. For example, every element in the top row (the first period) has one orbital for its electrons. As you move down the table, every row adds an orbital. Besides, what do the rows mean on a periodic table?
The rows on the periodic table are called periods. All the elements in a period have valence electrons in the same shell. The number of valence electrons increases from left to right in the period. When the shell is full, a new row is started and the process repeats.
Furthermore, what is a period number on the periodic table? A period number is the number that is given to a group of elements across the periodic table that have made a round from completing its outer electron shell. This pattern usually begins with a Group I element and ending with a Group 8 element.
Thereof, what does group number in periodic table mean?
The group number in the periodic table represents number of valence electrons of the elements in a certain group. For example, all the elements in Group−1 have 1 electron in their outer most shell. Answer link.
What is the significance of horizontal rows on the periodic table?
elements with similar properties are placed in the same vertical columns. vary from left to right along the rows. An unknown element is shiny and is found to be a good conductor of electricity.
Related Question Answers
Why are rows called periods?
The rows on the periodic table are called periods. All the elements in a period have valence electrons in the same shell. The number of valence electrons increases from left to right in the period. When the shell is full, a new row is started and the process repeats. What is the difference between rows and columns in the periodic table?
Periods are horizontal rows (across) the periodic table, while groups are vertical columns (down) the table. Atomic number increases as you move down a group or across a period. What is group and period in periodic table?
Groups and periods are two ways of categorizing elements in the periodic table. Periods are horizontal rows (across) the periodic table, while groups are vertical columns (down) the table. Atomic number increases as you move down a group or across a period. Where are the transition metals?
The transition metals are a group of metals that are found in the middle of the periodic table. The alkaline earth metals, beginning with beryllium are to the left and the boron group elements are to the right. How many row and groups are there in periodic table?
⚡? The Modern Periodic Table has eighteen vertical columns and seven horizontal rows. ⚡? The eighteen vertical columns are termed as ''GROUPS" & the seven horizontal rows are termed as ''PERIODS". How are rows in the periodic table organized?
The periodic table of elements arranges all of the known chemical elements in an informative array. Elements are arranged from left to right and top to bottom in order of increasing atomic number. Order generally coincides with increasing atomic mass. The rows are called periods. What are the 7 families of the periodic table?
FAMILIES OF ELEMENTS: Related elements, including the noble gases, halogens, alkali metals, alkaline earth metals, transition metals, lanthanides, and actinides. In addition, metals, nonmetals, and metalloids form loosely defined families. Other family designations—such as carbon family—are sometimes used. What are the numbers on the top of the periodic table?
The number above the symbol is the atomic mass (or atomic weight). This is the total number of protons and neutrons in an atom. The number below the symbol is the atomic number and this reflects the number of protons in the nucleus of each element's atom. How many groups are there in periodic table?
18
What is Group 1 on the periodic table called?
Group 1 - the alkali metals. The Group 1 elements in the periodic table are known as the alkali metals. They include lithium, sodium and potassium, which all react vigorously with water to produce an alkaline solution. What is Group 12 on the periodic table called?
Group 12, by modern IUPAC numbering, is a group of chemical elements in the periodic table. It includes zinc (Zn), cadmium (Cd) and mercury (Hg). Formerly this group was named IIB (pronounced as "group two B", as the "II" is a Roman numeral) by CAS and old IUPAC system. What is period table?
The periodic table, also known as the periodic table of elements, is a tabular display of the chemical elements, which are arranged by atomic number, electron configuration, and recurring chemical properties. The seven rows of the table, called periods, generally have metals on the left and nonmetals on the right. What is Group 3 on the periodic table called?
The Transition metals. The 38 elements in groups 3 through 12 of the periodic table are called "transition metals." As with all metals, the transition elements are both ductile and malleable, and conduct electricity and heat. What are the horizontal rows known as?
Horizontal rows in periodic table are called periods , they are seven in numbers elements in same period may not have similar quality. while vertical rows in periodic table are called columns or family. What do the rows and columns represent on the periodic table?
The vertical columns on the periodic table are called groups or families because of their similar chemical behavior. All the members of a family of elements have the same number of valence electrons and similar chemical properties. The horizontal rows on the periodic table are called periods. How is the chemical symbol of an element determined?
The number of protons defines what element it is and also determines the chemical behavior of the element. Atomic symbol: The atomic symbol (or element symbol) is an abbreviation chosen to represent an element ("C" for carbon, "H" for hydrogen and "O" for oxygen, etc.). What are the 8 families of the periodic table?
FAMILIES OF ELEMENTS: Related elements, including the noble gases, halogens, alkali metals, alkaline earth metals, transition metals, lanthanides, and actinides. In addition, metals, nonmetals, and metalloids form loosely defined families. Other family designations—such as carbon family—are sometimes used.