news /
What is the chemical formula and Colour of Mohr's salt crystals?
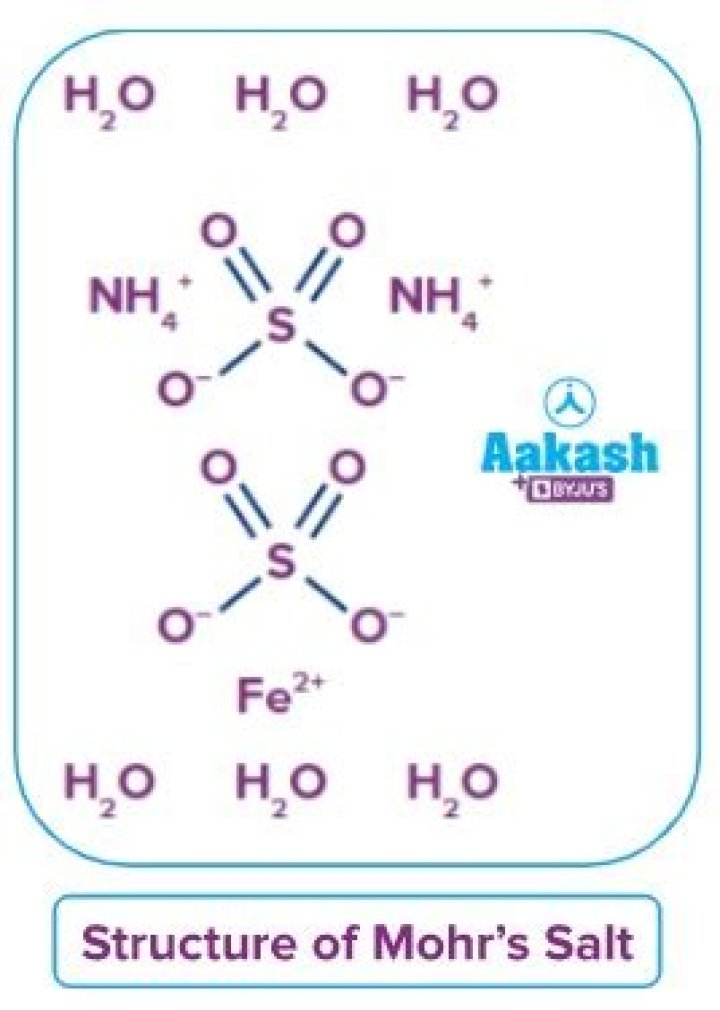
Ammonium iron(II) sulfate
| Names | |
|---|---|
| Chemical formula | Fe(SO4)(NH4)2(SO4) (anhydrous) Fe(SO4)(NH4)2(SO4)·6H2O (hexahydrate) |
| Molar mass | 284.05 g mol−1 (anhydrous) 392.13 g mol−1 (hexahydrate) |
| Appearance | Blue-green solid |
| Density | 1.86 g/cm3 |
Also, what is the formula of Mohr's salt?
(NH4)2Fe(SO4)2·6H2O
Furthermore, what is the other name of Mohr salt? Ammonium iron(II) sulfate, or Mohr's salt, is the inorganic compound with the formula (NH4)2Fe(SO4)2(H2O)6.
Similarly, what is the Colour of ferrous ammonium sulphate?
light green
What is the chemical formula of iron sulphate?
FeSO4
Related Question Answers
Is Mohr's salt acidic or basic?
The ammonium ions make solutions of Mohr's salt slightly acidic, which slows this oxidation process. Sulfuric acid is commonly added to solutions to reduce oxidation to ferric iron.What is the Colour of Mohr salt?
Ferrous Ammonium Sulphate is also called Mohr's salt a light green color sand like crystalline solid. It is a hazardous substance and preparation of ferrous ammonium sulfate is carried out from equimolar mixture of hydrated ferrous sulphate and ammonium sulphate in water; containing a little of sulphuric acid.What is Mohr's salt used for?
Mohr's salt is used in analytical chemistry as the preferred source of ferrous ions due to the solid's extended shelf life and resistance to oxidation. Sulfuric acid is often added to solutions using ferrous ammonium sulfate in order to reduce the oxidation of the ferric iron.Why do we add H2SO4 to Mohr's salt?
Ferrous ions of Mohr's salt undergo hydrolysis in aqueous solution. to prevent the hydrolysis, Conc. H2SO4 needs to be added to the Mohr's salt crystals during the preparation of its standard solution. Another reason is also to prevent the oxidation of Fe2+ ions of Mohr's salt to Fe3+ions.What is the meaning of double salt?
A double salt is a salt that contains more than one cation or more than one anion. Other examples include potassium sodium tartrate, ammonium iron(II) sulfate (Mohr's salt), and bromlite. The fluorocarbonates contain fluoride and carbonate anions. Many coordination complexes form double salts.What is the N factor of Mohr salt?
In Mohr's salt Fe is in +2 state (Fe2 +) , it oxidises by removing 1 electron and converts to Fe3 +. Therefore n- factor = 1. ( 1 electron is removed per mole of Mohr's salt).What do you mean by more salt?
: a light green crystalline salt FeSO4.(NH4)2SO4.6H2O used chiefly in iron plating, in photography, and in chemical analysis; ferrous ammonium sulfate.Is Mohr's salt poisonous?
HAZARD SUMMARY* Ferrous Ammonium Sulfate can affect you when breathed in. * Contact can irritate the skin and eyes. * Breathing Ferrous Ammonium Sulfate can irritate the nose and throat causing coughing and wheezing. * High exposure may cause nausea, stomach pain, diarrhea, vomiting and drowsiness.
How can we prepare 0.1 N feso4 solution?
Preparation and Standardization of 0.1 M Ferrous Ammonium Sulphate- Dissolve 40 g of ferrous ammonium sulfate in a previously cooled mixture of 40 ml of sulphuric acid and 200 ml of water.
- Dilute with sufficient freshly boiled and cooled water to produce 1000 ml.
- Standardize the solution in the following manner.
What is Iron III alum?
Double sulfate of Iron (III) and ammonium is also known as ferric alum. Here, "alum" points to the wide class of double salts of similar composition: MIMII(SO4)2·12H2O, where MI is an univalent ion, usually K or NH4, and MII is trivalent: Al, Fe, Cr.What will happen if we drink Ferrous ammonium sulphate?
SOLID Irritating to skin and eyes. If swallowed will cause nausea and vomiting.Why FAS is used in titration?
After all the organic compounds are oxidized, some dichromate electron acceptor remains and it is this remaining amount that is measured in the titration with the FAS. The ferrous (Fe2+) in FAS donates electrons to the remaining dichromate and is oxidized to ferric (Fe3+) while the dichromate is reduced.What kind of reagent is iron alum?
Ammonium iron(III) sulfate, NH4Fe(SO4)2·12 H2O, or NH4[Fe(H2O)6](SO4)2·6 H2O, also known as ferric ammonium sulfate (FAS) or iron alum, is a double salt in the class of alums, which consists of compounds with the general formula AB(SO4)2 · 12 H2O.Is tutton salt soluble in water?
All Tutton's salts are highly or moderately soluble in water and have monoclinic crystal system.What is ferric alum?
Ferric alum is a less pure form of aluminum sulphate. It is used in lower grades of paper for loading and sizing purposes. Ferric alum uses bauxite as the raw material. The raw material is cooked with sulphuric acid under suitable conditions. The resultant product is sold in liquid form.Why is ferrous sulphate called hydrated salt?
The hydrated form is used medically to treat iron deficiency, and also for industrial applications. The name copperas dates from times when the copper(II) sulfate was known as blue copperas, and perhaps in analogy, iron(II) and zinc sulfate were known respectively as green and white copperas.How do you make a ferric indicator?
Procedure. Dissolve 50 g of ferric ammonium sulfate R in a mixture of 300 mL of water and 6 mL of sulfuric acid (~1760 g/L) TS. Dilute with sufficient water to produce 1000 mL. Method of standardization.How do you do a titration of Mohr's salt?
- Fill the burette by potassium permanganate solution.
- Take conical flask and add 5ml of dilute sulfuric acid in it.
- Pipette out 10 ml of prepared standard Mohr's salt solution in the same conical flask.
- Place a white tile under the burette and place the conical flask containing Mohr's salt solution and H2SO4 on it.
What are the two different salts of Mohar?
Mohr's salt is an inorganic compound which is a double salt with the formula (NH4)2Fe(SO4)2(H2O)6. It is a common laboratory reagent with IUPAC name ammonium iron(II) sulfate.Observation Table:
| Color of the crystals | Light Green |
|---|---|
| Solubility in water | Soluble |
| Blue litmus paper (Litmus test) | Blue litmus paper turns red |