What type of reaction is CA O2?
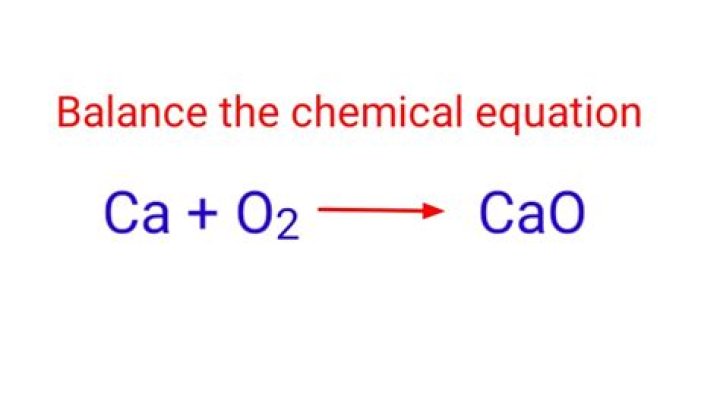
Beside this, what type of reaction is calcium and oxygen?
A great many common reactions are exothermic. The formation of compounds from the constituent elements is almost always exothermic. Formation of water from molecular hydrogen and oxygen and the formation of a metal oxide such as calcium oxide (CaO) from calcium metal and oxygen gas are examples.
One may also ask, is CA O2 a combustion reaction? 2Ca + O2. 2CaO
Сalcium react with oxygen to produce calcium oxide. This reaction takes place at a temperature of over 300°C. Calcium combustion in air.
Simply so, what type of reaction is O2?
Combustion reactions
What type of reaction is the equation C O2 → CO2?
Another special type of combination reaction is a combustion, meaning a reaction in which something combines with oxygen and gives off heat and light: C + O2 → CO2 2 Mg + O2 → 2 MgO Combustion of a compound includes break-up (decomposition) of the compound first.
Related Question Answers
What happens when Ca reacts with O2?
The metal reacts slowly with oxygen, water vapour, and nitrogen of the air to form a yellow coating of the oxide, hydroxide, and nitride. It burns in air or pure oxygen to form the oxide and reacts rapidly with warm water (and more slowly with cold water) to produce hydrogen gas and calcium hydroxide.What happens when Aluminium reacts with oxygen?
Aluminum also reacts with oxygen to form aluminum oxide.Is calcium stored in kerosene?
Calcium does not gives a violent reaction to air and hence, not stored in kerosene.Does copper react with oxygen?
For example, copper and oxygen react together to make copper oxide. Copper and oxygen are the reactants because they are on the left of the arrow. You can see that now there are two copper atoms and two oxygen atoms on each side. This matches what happens in the reaction.Does Zinc react with oxygen?
Zinc reacts with oxygen in moist air. The metal burns in air to form zinc(II) oxide, a material that goes from white to yellow on prolonged heating.Why Calcium is said to be oxidized in this reaction?
Because calcium has gained in oxidation number due to the loss of electrons, we say it has been oxidized. Oxygen on the other hand has decreased in oxidation number due to a gain of electrons so we say it has been reduced.Why is CaO not CaO2?
The Calcium ion is an Alkaline earth metal and wants to give up the 2 s orbital elections and become a +2 cation. Oxygen has six valence electrons and is looking to gain two electrons to complete the octet (8) electron count in the valence shell making it a -2 anion.Is calcium a good conductor of electricity?
While calcium is a poorer conductor of electricity than copper or aluminium by volume, it is a better conductor by mass than both due to its very low density.Is C O2 CO2 a exothermic reaction?
It is an exothermic reaction as heat is evolved during the reaction. It is a combination reaction as carbon combines with oxygen to form carbon dioxide. It is a combustion reaction as carbon is burnt in the presence of oxygen.What type of reaction is kclo3 KCL O2?
The thermal decomposition of potassium chlorate to produce potassium chloride and oxygen. This reaction takes place at a temperature of 150-300°C.What type of reaction is 2H2O 2H2 O2?
synthesis reactionIs CO2 endothermic or exothermic?
Answer. The formation of co2 is an Exothermic reaction in which heat is evolved.Is C O2 CO2 balanced?
Carbon monoxide reacts with oxygen to produce carbon dioxide. Write the balanced chemical equation for this reaction.Example.
| Step | Result | Equation balanced? |
|---|---|---|
| 1 | carbon monoxide + oxygen → carbon dioxide | |
| 2 | CO + O 2 → CO 2 | |
| 3 | Reactants: 1 × C, (1 × O) + (2 × O) = 3 × O. Products: 1 × C, 2 × O | Not balanced. |
| 4 | 2CO + O 2 → CO 2 |