Why didn't Scientists know much about atoms?
Simply so, what is the significance of the Periodic Table of Elements choose the best answer?
Choose the best answer. It lists all the different metals known to humans. It predicts and lists all the chemical elements in the universe.
Also, who proved the existence of atoms? Einstein
Secondly, how certain are scientists about the structure of the atom?
Mr. Roentgen's x-rays allowed scientists to measure the size of the atom. The x-rays were small enough to discern the atomic clouds. This was done by scattering x-rays from atoms and measuring their size just as Rutherford had done earlier by hitting atoms with other nuclei starting with alpha particles.
Why is it useful to model an atom?
Atomic models are important because, they help us visualize the interior of atoms and molecules, and thereby predicting properties of matter.
Related Question Answers
Do atoms contain empty space?
In reality, atoms do not contain any empty space. Rather, they are filled completely with spread-out electrons, making the shrinking of atoms impossible.Why can't Scientists see atoms?
Almost all of an atom's mass comes from the protons and neutrons in the nucleus. However, because electrons orbit around the nucleus, most of an atom is empty space! You can't see atoms with the naked eye, because they're simply too small. Using electron microscopes, scientists have been able to study atoms.What element has 4shells?
BerylliumWhy are they called atoms?
The term "atom" comes from the Greek word for indivisible, because it was once thought that atoms were the smallest things in the universe and could not be divided. We now know that atoms are made up of three particles: protons, neutrons and electrons — which are composed of even smaller particles, such as quarks.How is the chemical symbol of an element determined?
The number of protons defines what element it is and also determines the chemical behavior of the element. Atomic symbol: The atomic symbol (or element symbol) is an abbreviation chosen to represent an element ("C" for carbon, "H" for hydrogen and "O" for oxygen, etc.). The other two types are protons and neutrons.What does the number 12 represent on the periodic table?
The Elements, sorted by Atomic Number| Atomic Number | Symbol | Name |
|---|---|---|
| 9 | F | Fluorine |
| 10 | Ne | Neon |
| 11 | Na | Sodium |
| 12 | Mg | Magnesium |
What is the center of an atom called?
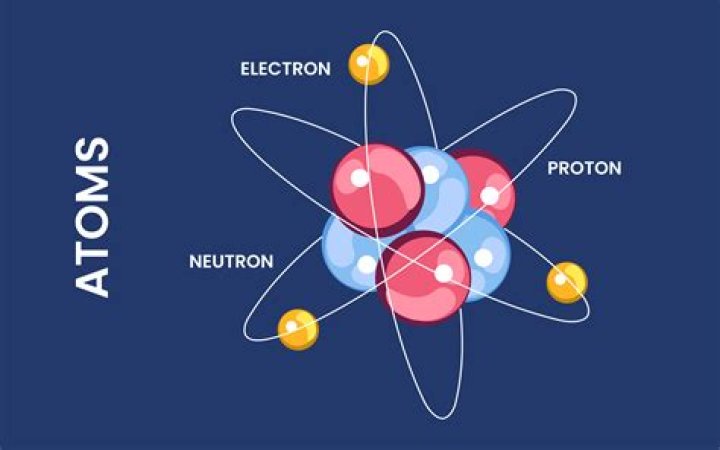
Atoms consist of three basic particles: protons, electrons, and neutrons. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). The outermost regions of the atom are called electron shells and contain the electrons (negatively charged).What has a positive charge in an atom?
Atoms are made of extremely tiny particles called protons, neutrons, and electrons. Protons and neutrons are in the center of the atom, making up the nucleus. Electrons surround the nucleus. Protons have a positive charge.Can scientists see an atom?
Atoms are really small. So small, in fact, that it's impossible to see one with the naked eye, even with the most powerful of microscopes. Now, a photograph shows a single atom floating in an electric field, and it's large enough to see without any kind of microscope. ?? Science is badass.Do atoms die?
Since an atom has a finite number of protons and neutrons, it will generally emit particles until it gets to a point where its half-life is so long, it is effectively stable. It undergoes something known as “alpha decay,” and it's half-life is over a billion times longer than the current estimated age of the universe.Can atoms be destroyed?
All matter consists of indivisible particles called atoms. Atoms of the same element are similar in shape and mass, but differ from the atoms of other elements. Atoms cannot be created or destroyed.How did Einstein prove atoms existed?
Einstein's theory was that that the particles from the pollen grains were being moved around because they were constantly crashing into millions of tinier molecules of water – molecules that were made of atoms.How do atoms look?
It turns out that visible light is no good for seeing the shape of atoms, because its wavelength is way bigger than the atom. So, while individual atoms can reflect visible light, because atoms are so much smaller than the light itself, you will never be able to see any more than a fuzzy dot.Are humans made of atoms?
About 99 percent of your body is made up of atoms of hydrogen, carbon, nitrogen and oxygen. You also contain much smaller amounts of the other elements that are essential for life. The hydrogen atoms in you were produced in the big bang, and the carbon, nitrogen and oxygen atoms were made in burning stars.Is there anything smaller than an atom?
In the physical sciences, subatomic particles are smaller than atoms. They can be composite particles, such as the neutron and proton; or elementary particles, which according to the standard model are not made of other particles.How much of matter is empty space?
A hydrogen atom is about 99.9999999999996% empty space. Put another way, if a hydrogen atom were the size of the earth, the proton at its center would be about 200 meters (600 feet) across.Do atoms touch?
If "touching" is taken to mean that two atoms influence each other significantly, then atoms do indeed touch, but only when they get close enough. With 95% of the atom's electron probability density contained in this mathematical surface, we could say that atoms do not touch until their 95% regions begin to overlap.Who discovered atom first?
DemocritusIs there a real picture of an atom?
Physicist: Actual pictures of atoms aren't actually pictures at all. But the wavelength of visible light is about half a micrometer (a two-millionth of a meter) and atoms are around one ångström (a ten-billionth of a meter) across. On the scale of atoms, visible light acts too wonky to be used for photographs.How long have we known about atoms?
Around 450 B.C., the Greek philosopher Democritus introduced the idea of the atom. However, the idea was essentially forgotten for more than 2000 years. In 1800, John Dalton re-introduced the atom. He provided evidence for atoms and developed atomic theory.Are we mostly empty space?
But it might humble you to know that all of those things – your friends, your office, your really big car, you yourself, and even everything in this incredible, vast Universe – are almost entirely, 99.9999999 percent empty space.Is Air made out of atoms?
The Air We Breathe.Air is actually a collection of very small molecules; oxygen gas and nitrogen gas are molecules made from two atoms each. Only the noble gases readily exist as single atoms.